|
3/14/2023 0 Comments Nitrogen atomLegumes like peas, clover and peanuts have little nodules on their roots that attract bacteria that convert stubborn atmospheric nitrogen into ammonia or ammonium, which can then be used to power the plant. On the other end of the spectrum, the most common way nitrogen is made available to organisms is when atmospheric nitrogen is fixed by bacteria, some of which live free in the soil and others of which enjoy a symbiotic relationship with certain plant species. This falls to the ground as rain, where plants slurp it up and use it for their biological processes. If you've ever wondered why your outdoor plants seem happier after a rain than they do when you turn a sprinkler on them, there's a reason for that: Lightning electrifies atmospheric nitrogen (N2) and water (H2O) to reconfigure them into ammonia (NH3) and nitrates (NO3). At room temperatures, nitrogen dioxide is a reddish-brown gas that has a density of 1.8 g/dm 3. Alkaloid names generally end in the suffix. Nitrogen Dioxide (NO 2) is a covalent compound that is composed of a central nitrogen atom single bonded to an oxygen atom and a double bond with another oxygen atom. Ball-and-stick model, geometric structure. Nitrogen oxide, nitrogen monoxide or Oxidonitrogen. Most alkaloids have one or more of their nitrogen atoms as part of a ring of atoms, frequently called a cyclic system. RF 2AMTCRKNitric oxide, NO, molecule model and chemical formula. 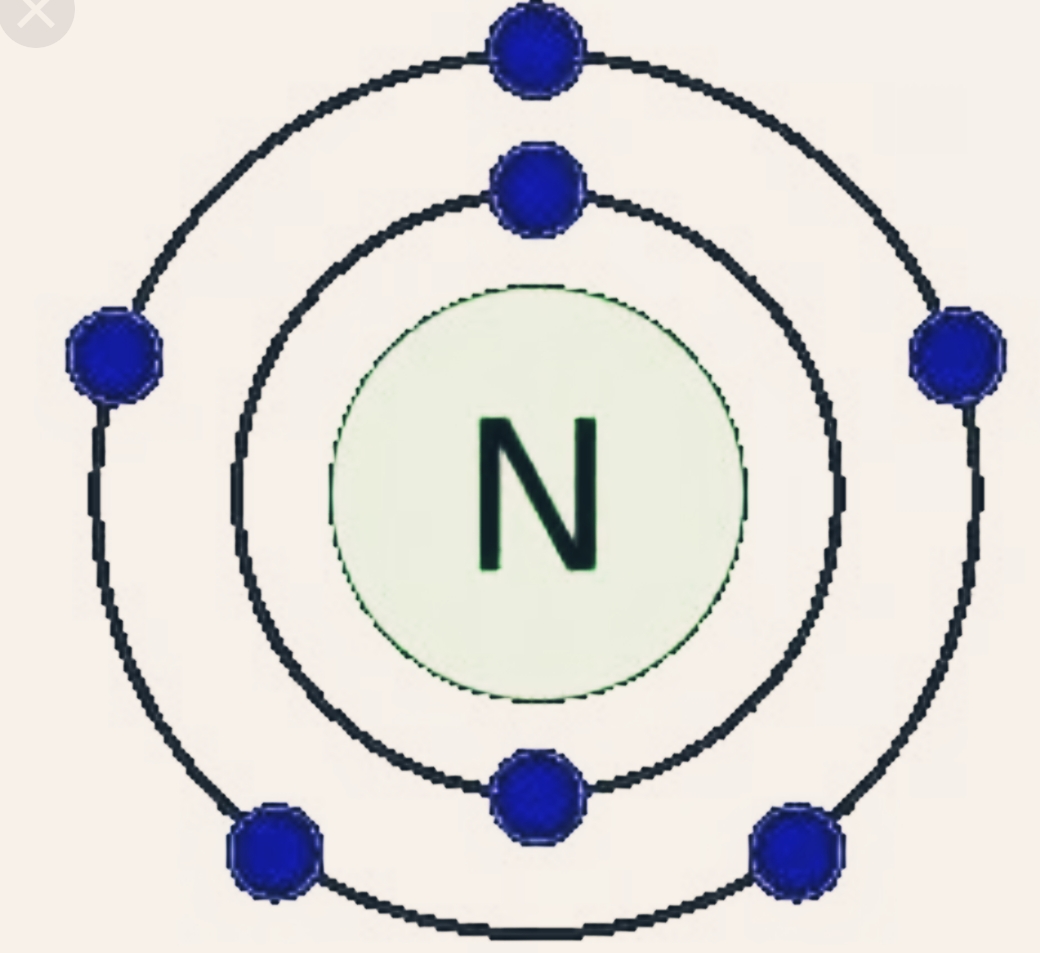
The name alkaloid (alkali-like) was originally applied to the substances because, like the inorganic alkalis, they react with acids to form salts. Atmospheric nitrogen (N2) is very stable, so it takes an incredible amount of energy to convert it to a different form. This or another nitrogen atom can be active as a base in acid-base reactions. Step 1: Nitrogen Fixationīelieve it or not, lightning and bacteria are primarily responsible for turning atmospheric nitrogen into nitrogen living things can use. Nitrogen atoms are ubiquitous in biologically significant secondary metabolites (alkaloids, cytokinins), biomacromolecules (proteins, peptides, DNA, RNA) as. 
This is the cycle of a nitrogen atom on Earth, and its journey starts either very quietly or with a humongous bang. Since nitrogen is a limited resource on this planet, a nitrogen atom doesn't spend much time doing nothing when it's in a form living things can use - scientists call this nitrogen "fixed." Fixed nitrogen is taken up by plants, which are eaten by animals, which eat other animals, which die and decompose and release nitrogen back into the ecosystem to be worked on by bacteria or plants.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
